hek293 ptet off advanced cell line (TaKaRa)
Structured Review

Hek293 Ptet Off Advanced Cell Line, supplied by TaKaRa, used in various techniques. Bioz Stars score: 94/100, based on 207 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more
https://www.bioz.com/result/hek293 ptet off advanced cell line/product/TaKaRa
Average 94 stars, based on 207 article reviews
Images
1) Product Images from "Development of Novel Epigenetic Anti-Cancer Therapy Targeting TET Proteins."
Article Title: Development of Novel Epigenetic Anti-Cancer Therapy Targeting TET Proteins.
Journal: International journal of molecular sciences
doi: 10.3390/ijms242216375

Figure Legend Snippet: Figure 1. Establishment of the TET agonist screening system. (a) Inducible expression of the catalytic domain (CD) of TET1 was achieved by stable transfection of FLAG-HA (FH) tagged as TET1-CD in the pTRE-Tight vector into HEK293 Tet-Off cells. FH-TET1-CD expression was triggered upon the
Techniques Used: Expressing, Stable Transfection, Plasmid Preparation
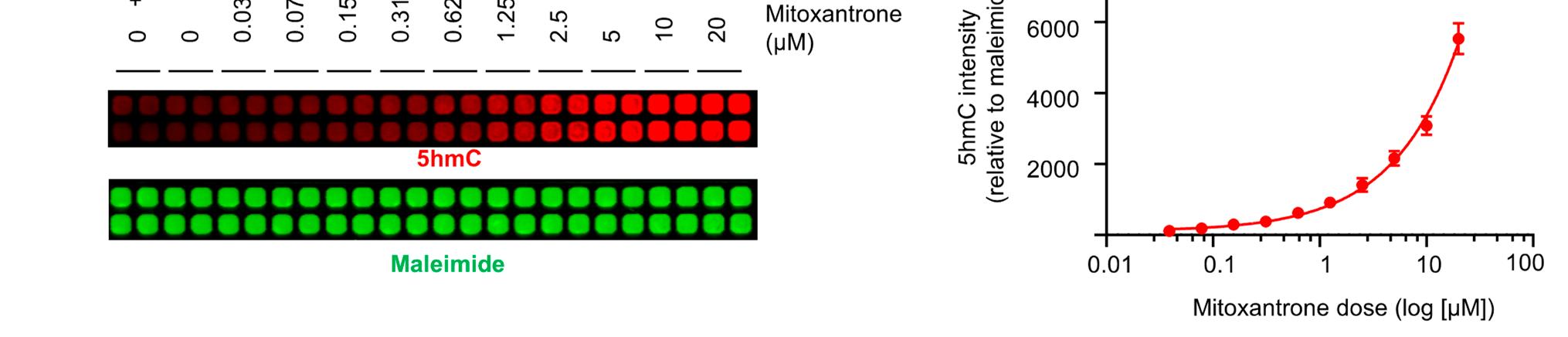
Figure Legend Snippet: Figure 2. Identification of mitoxantrone as a potential TET agonist through primary screening. (a) HEK#15 cells (4 × 104 cells per well in 50 µL) cultured with (+) or without (−) Dox were seeded and maintained in a 384-well plate. After 2 days, the plate was co-stained with antibodies specific to HA (green) and 5hmC (red). Maleimide staining was used to monitor compound-induced cytotoxicity. (b,c) Levels of FH-TET1-CD protein and 5hmC in individual wells of the 384-well plate described in (a). Uniform staining intensities for TET1-CD and 5hmC within individual wells were observed. The Z’ factor values are also provided. Signal intensities were normalized to the mean of the maleimide staining intensities. (d) Summary of the results presented in (b,c). The mean ± s.d. is shown. (e) Molecular structure of NSC 279836, also known as mitoxantrone. (f) Dose-dependent elevation in 5hmC intensities in HEK15 cells upon mitoxantrone treatment. Maleimide staining was used to monitor compound-induced cytotoxicity. (g) Summary of the 5hmC signal intensities shown in (f). The 5hmC signal intensity was calculated by subtracting the mean of the vehicle-treated group from that of the drug-treated groups, which was then normalized to the mean of maleimide staining intensities. All data are presented as the mean ± s.d. The p-values were determined by unpaired Student’s t-test. **** p ≤0.0001.
Techniques Used: Cell Culture, Staining
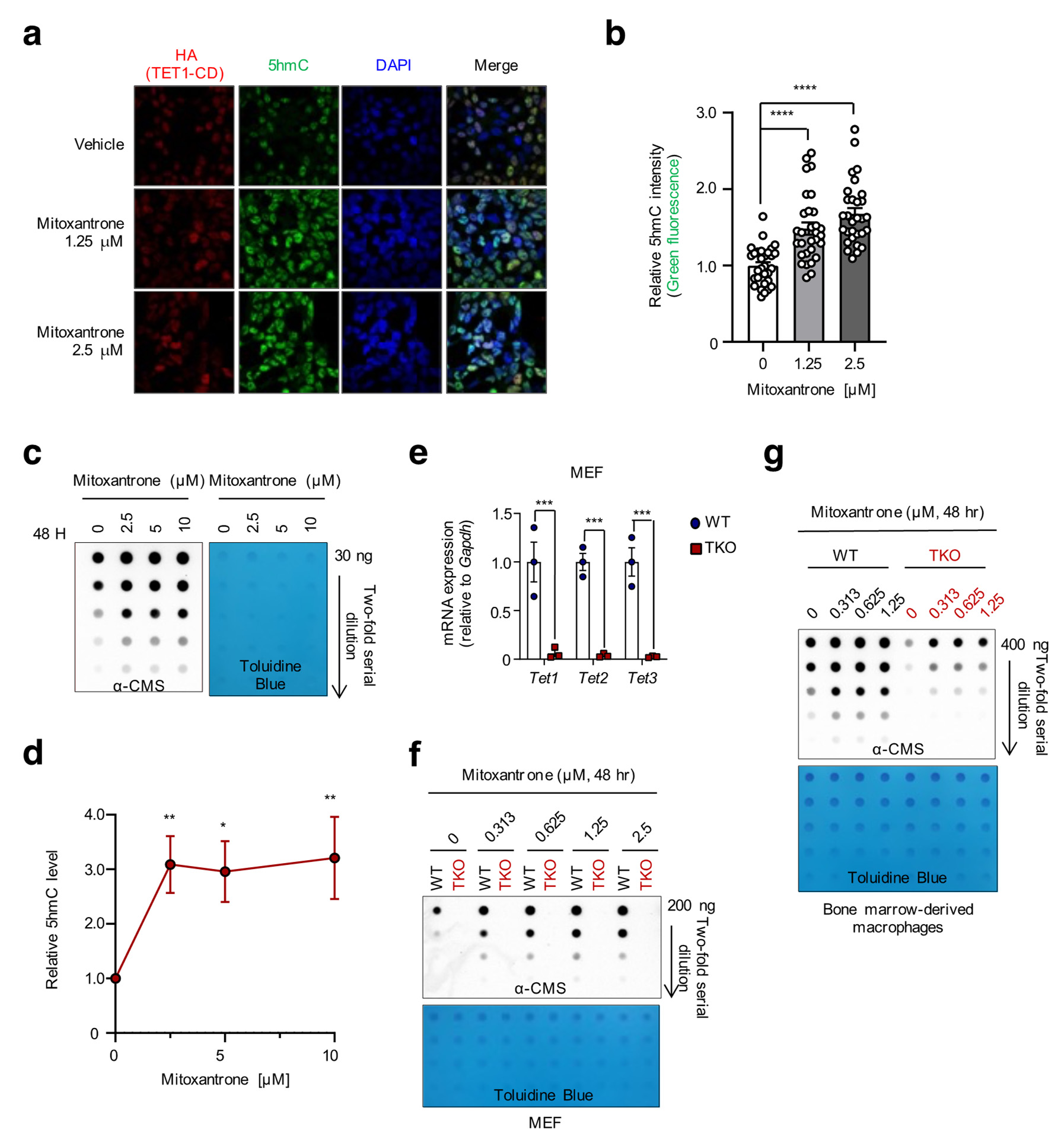
Figure Legend Snippet: Figure 3. Mitoxantrone augments 5hmC levels in a TET-dependent manner. (a) Immunocytochem- istry revealed the mitoxantrone-induced increase in 5hmC levels. HEK#15 cells, cultured without Dox, were treated with mitoxantrone at the indicated concentration and co-stained with antibodies specific for the HA epitope (red) and 5hmC (green). DAPI (blue) indicated nuclear staining. (b) Quantification of 5hmC intensities shown in (a). (c) Genomic DNA purified from HEK#15 cells treated with mitox- antrone as indicated was treated with bisulfite to convert 5hmC to cytosine 5-methylenesulfonate (CMS). CMS was quantified by dot blot assay using an anti-CMS antibody. Toluidine blue staining was used to monitor equivalent DNA loading. (d) Summary of the 5hmC intensities shown in (c). (e) Quantitative RT-PCR was performed to assess the levels of Tet1, Tet2, and Tet3 mRNAs relative to Gapdh in WT and TET triple knockout (TKO) MEFs (n = 3). (f,g) WT and TET TKO MEFs (f) or BMMs (f) were treated with increasing concentrations of mitoxantrone for 48 h, followed by quantification of 5hmC by anti-CMS dot blot. Toluidine blue staining was used to monitor equivalent DNA loading. All data are presented as the mean ± s.d. The p-values were determined by unpaired Student’s t-test. * p ≤0.05, ** p ≤0.01, *** p ≤0.001, and **** p ≤0.0001.
Techniques Used: Cell Culture, Concentration Assay, Staining, Dot Blot, Quantitative RT-PCR, Triple Knockout